|
Many elements exist as solid-state at room temperatures, such as sodium, vanadium and magnesium. is made up of a unique arrangement of atoms. In case of rubber constituent particles are not arranged in a perfectly ordered manner so it is not a crystalline solid, as it is amorphous solid. 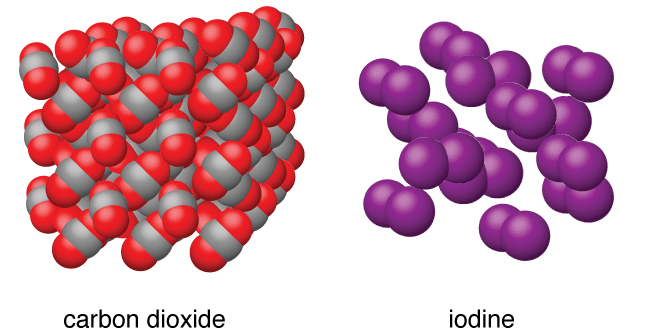
The pressure exerted by a vapor in equilibrium with its corresponding liquid at a given temperature is called its a. Describe the difference between crystalline and amorphous solids. Crystalline solids are true solids, have a regular arrangement of particles (long range order) whereas amorphous solids have an irregular arrangement of. In other words, you can simply say that crystalline solid consists of particles that are arranged in a 3-dimensional manner. They can be crystalline, like table salt, or amorphous, like glass, rubber or plastic. Particles in a(n) solid are arranged in an orderly repeating pattern in space. According to the definition of crystalline solid, a material whose molecules, atoms, or even sub-atomic particles are arranged in a highly ordered structure is known as crystalline solids. Solids are incompressible and have high density, compared to liquids and gases. The particles vibrate back and forth within their fixed positions and do not move freely. As a result, solids have a definite shape and volume. In the solid state, the particles do not have enough energy to overcome the strong intermolecular forces, which means they are tightly held against each other. The main types of crystalline solids are ionic solids, metallic solids, covalent network solids, and molecular solids. The particles in an amorphic solid are arranged in a somewhat random hap hazard manner. Some substances form crystalline solids consisting of particles in a very organized structure others form amorphous (noncrystalline) solids with an internal structure that is not ordered. But amorphous solids often consist of large particles that are not. The particles (atoms, molecules or ions) in a crystalline solid are arranged in a regular repeating geometrical pattern in 3-Dimensional space. The particles in crystalline solids are arranged in a repeating, three- dimensional pattern called a crystal. Viscosity – resistance to motion that exists between the molecules of a liquid when they move past each other Properties of Solids Solids can be classified into two broad categories crystalline solids and amorphic solids (or non-crystalline) solids. Surface tension – force that pulls adjacent parts of a liquid’s surface together, decreasing the surface area to the smallest size possible 
Intermolecular forces – forces of attraction or repulsion existing between neighboring particles. Solids, liquids and gases are all around us, they are the three main states of matter – but how many of their properties do you really know? Let’s find out! Topics Covered in Other ArticlesĬapillary action – attraction of the surface of a liquid to the surface of a soliĬrystalline – the particles of the solid are arranged in an orderly, geometric, repeating patternĭiffusion – spontaneous mixing of the particles of two substances caused by their motion In this tutorial, you will learn about the properties of the solid, liquid, and gas phases of matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
